Sarepta Therapeutics, Inc. Aktie News
Sarepta Therapeutics, Inc. Aktie News

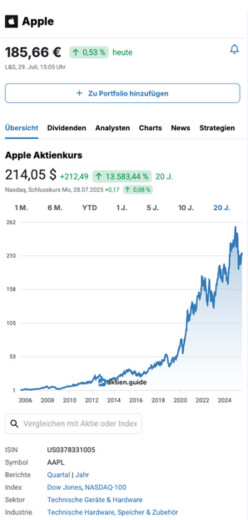
15,46 €
1,23 %
heute
L&S,
9. Februar, 15:30 Uhr
Index
Industrie
Sarepta Therapeutics, Inc. Aktie News
CAMBRIDGE, Mass.--(BUSINESS WIRE)--Sarepta Therapeutics, Inc. (NASDAQ:SRPT), the leader in precision genetic medicine for rare diseases, today announced that Medsafe, the New Zealand Medicines and Medical Devices Safety Authority, has granted approval for its clinical trial application (CTA) for Study SRP-1005-101, also known as INSIGHTT. Sarepta expects to initiate this first-in-human clinical...
Sarepta Therapeutics, Inc. (SRPT) Discusses 3-Year Topline Results From EMBARK Phase 3 Trial of ELEVIDYS in Duchenne Muscular Dystrophy Transcript
Sarepta Therapeutics Inc (NASDAQ:SRPT) shares moved higher in early trade on Monday as it reported positive topline three-year results from its Phase 3 EMBARK study evaluating its gene therapy ELEVIDYS in ambulatory patients with Duchenne muscular dystrophy. Sarepta said patients treated with ELEVIDYS demonstrated statistically significant and clinically meaningful slowing of disease progressio...
Sarepta stock jumped early Monday after the company unveiled three-year test results for its embattled gene therapy, Elevidys.
Sarepta reports positive, statistically significant results in a study evaluating the efficacy of its blockbuster gene therapy.
Sarepta Therapeutics' gene therapy for a progressive muscle‑wasting disorder showed sustained benefit in patients' ability to control and coordinate movement, according to three‑year data from a late‑stage study released on Monday.
CAMBRIDGE, Mass.--(BUSINESS WIRE)--Sarepta Therapeutics, Inc. (NASDAQ:SRPT), the leader in precision genetic medicine for rare diseases, today announced positive topline three-year functional results from Part 1-treated patients in EMBARK (Study SRP-9001-301), the global, randomized placebo-controlled Phase 3 study evaluating ELEVIDYS (delandistrogene moxeparvovec-rokl) in ambulatory individual...
CAMBRIDGE, Mass.--(BUSINESS WIRE)--Sarepta Therapeutics, Inc. (NASDAQ:SRPT), the leader in precision genetic medicine for rare diseases, today announced that on Monday, Jan. 26, 2026, at 8:30 am Eastern Time, the Company will host a webcast and conference call to present 3-year topline functional results from patients treated in Part 1 of EMBARK (Study 9001-301), the global, randomized placebo-...
Kostenlos registrieren
aktien.guide ist das Tool zum einfachen Finden, Analysieren und Beobachten von Aktien. Lerne von erfolgreichen Investoren und triff fundierte Anlageentscheidungen. Wir machen Dich zum selbstbestimmten Investor.