Celcuity Inc. Aktie News
Celcuity Inc. Aktie News

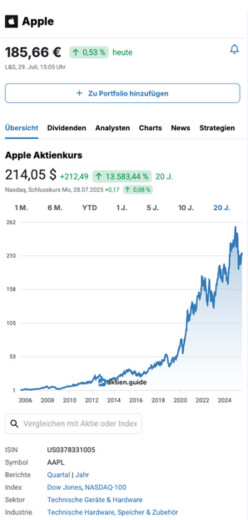
106,75 $
2,37 %
vorgestern
Nasdaq,
6. Februar, 22:00 Uhr
Celcuity Inc. Aktie News
David Dalvey sold 20,000 shares indirectly on Jan. 27, 2026 for a transaction value of ~$2.4 million, at a weighted average price of $120.03 per share. The transaction represented 18.18% of Dalvey's total indirect holdings prior to the sale, reducing his stake from 110,000 to 90,000 shares.
MINNEAPOLIS, Feb. 04, 2026 (GLOBE NEWSWIRE) -- Celcuity Inc. (Nasdaq: CELC), a clinical-stage biotechnology company pursuing development of targeted therapies for oncology, today announced its participation in the Guggenheim Emerging Outlook: Biotech Summit 2026 to be held in New York, New York on February 11-12, 2026.
FDA grants Priority Review and assigns a PDUFA goal date of July 17, 2026 MINNEAPOLIS, Jan. 20, 2026 (GLOBE NEWSWIRE) -- Celcuity Inc. (Nasdaq: CELC), a clinical-stage biotechnology company pursuing development of targeted therapies for oncology, today announced that the U.S. Food and Drug Administration (“FDA”) has accepted for filing its New Drug Application (“NDA”) for gedatolisib in hormone...
Celcuity Inc. surged over 750% in 2025 on best-in-class Phase 3 data for gedatolisib in HR+/HER2- advanced breast cancer. GEDATOLISIB demonstrated superior efficacy and safety versus standards of care, with median PFS of 9.3 months in VIKTORIA-1 and favorable adverse event profile. Management projects peak revenues of $2.5–$3 billion for gedatolisib; current cash position is nearly $500 million...
MINNEAPOLIS, Dec. 11, 2025 (GLOBE NEWSWIRE) -- Celcuity Inc. (Nasdaq: CELC), a clinical-stage biotechnology company pursuing development of targeted therapies for oncology, today announced updated results from the randomized, Phase 3 VIKTORIA-1 trial for gedatolisib, a multi-target PI3K/AKT/mTOR (“PAM”) inhibitor, in adults with hormone receptor positive (“HR+”), human epidermal growth factor r...
Many investors have profited from the artificial intelligence (AI) trade in 2025. But there have been several up-and-coming stocks in other sectors that have posted impressive gains this year.
A top biotech fund just made a bold move while Celcuity trades near record highs—here's what they might see coming next.
New York City-based Apis Capital Advisors added 776,000 shares in CELC during the third quarter, establishing a position valued at $38.3 million. The new stake represents 8.5% of Apis' reportable assets under management at quarter-end.
Kostenlos registrieren
aktien.guide ist das Tool zum einfachen Finden, Analysieren und Beobachten von Aktien. Lerne von erfolgreichen Investoren und triff fundierte Anlageentscheidungen. Wir machen Dich zum selbstbestimmten Investor.