Day One Biopharmaceuticals Inc Aktie News
Day One Biopharmaceuticals Inc Aktie News
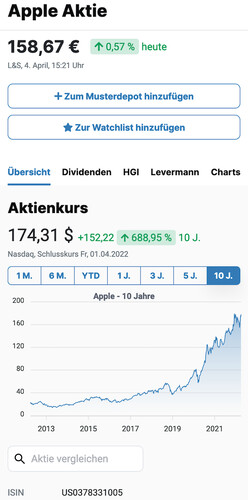
17,69 $
3,45 %
gestern
Nasdaq,
1. Mai, 22:00 Uhr
Day One Biopharmaceuticals Inc Aktie News
Neutral
GlobeNewsWire
●
8 Tage alt
Day One's OJEMDA™ (tovorafenib) Receives US FDA Accelerated Approval for Relapsed or Refractory BRAF-altered Pediatric Low-Grade Glioma (pLGG), the Most Co
Neutral
GlobeNewsWire
●
2 Monate alt
PDUFA target action date for tovorafenib NDA in relapsed or progressive pLGG remains set for April 30, 2024
Positiv
Seeking Alpha
●
3 Monate alt
Day One Biopharmaceuticals is a leader in pediatric oncology, with a focus on addressing the unmet needs of pediatric cancer patients. The company has a strong financial position, with enough cash to fund operations through 2026, and is progressing through clinical trials with promising results. Day One's lead product candidate, tovorafenib, has the potential to redefine the standard of care fo...
Neutral
GlobeNewsWire
●
4 Monate alt
BRISBANE, Calif., Dec. 19, 2023 (GLOBE NEWSWIRE) -- Day One Biopharmaceuticals (Nasdaq: DAWN) (“Day One” or the “Company”), a clinical-stage biopharmaceutical company dedicated to developing and commercializing targeted therapies for people of all ages with life-threatening diseases, today announced that Dr. Jeremy Bender, chief executive officer, will present during the 42nd Annual J.P. Morgan...
Neutral
GlobeNewsWire
●
6 Monate alt
Data subsets to be shared today in plenary oral presentations at the 2023 Society for Neuro-Oncology Annual Meeting Data subsets to be shared today in plenary oral presentations at the 2023 Society for Neuro-Oncology Annual Meeting
Neutral
GlobeNewsWire
●
6 Monate alt
NDA for tovorafenib in relapsed or progressive pLGG accepted for FDA priority review PDUFA target action date of April 30, 2024 BRISBANE, Calif., Nov. 06, 2023 (GLOBE NEWSWIRE) -- Day One Biopharmaceuticals (Nasdaq: DAWN) (“Day One” or the “Company”), a clinical-stage biopharmaceutical company dedicated to developing and commercializing targeted therapies for people of all ages with life-threat...
Neutral
GlobeNewsWire
●
6 Monate alt
BRISBANE, Calif., Oct. 30, 2023 (GLOBE NEWSWIRE) -- Day One Biopharmaceuticals (Nasdaq: DAWN) (“Day One” or the “Company”), a clinical-stage biopharmaceutical company dedicated to developing and commercializing targeted therapies for people of all ages with life-threatening diseases, today announced that the U.S. Food and Drug Administration (FDA) accepted its New Drug Application (NDA) for tov...
Positiv
Seeking Alpha
●
8 Monate alt
Day One Biopharmaceuticals, Inc. is awaiting an FDA filing decision on its lead drug tovorafenib for the treatment of pediatric low grade glioma in mid-November of 2023. The company is also running a phase 3 study to evaluate tovorafenib as a first-line treatment for pLGG. Tovorafenib is being explored alone and in combination with another drug in ongoing phase 1b/2 studies for MAPK pathway mut...
Kostenlos registrieren
aktien.guide ist das Tool zum einfachen Finden, Analysieren und Beobachten von Aktien. Lerne von erfolgreichen Investoren und triff fundierte Anlageentscheidungen. Wir machen Dich zum selbstbestimmten Investor.