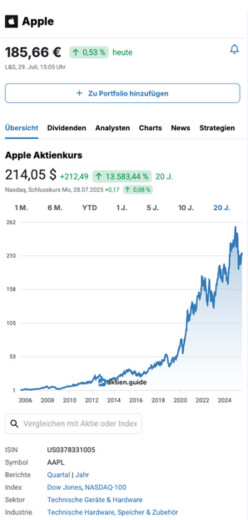
Vanda Pharmaceuticals Inc. Aktie News
Conference Call and Webcast to Follow WASHINGTON, Feb. 4, 2026 /PRNewswire/ -- Vanda Pharmaceuticals Inc. (Vanda) (Nasdaq: VNDA) today announced it will release results for the fourth quarter and full year 2025 on Wednesday, February 11, 2026, after the market closes. Vanda will host a conference call at 4:30 PM ET on Wednesday, February 11, 2026, during which management will discuss the fourth...
Vanda Pharmaceuticals Inc. (VNDA) Presents at 44th Annual J.P. Morgan Healthcare Conference Transcript
LONDON & NEW YORK--(BUSINESS WIRE)--This morning Vanda, a global leader of high-frequency positioning data and tactical macro insights and Exante Data Inc, an innovative data analytics and global macro strategy provider announced they completed a definitive merger agreement. Under the terms of the agreement, Exante Data Inc will merge with and into Vanda through the acquisition by Vanda of all ...
WASHINGTON, Jan. 8, 2026 /PRNewswire/ -- Vanda Pharmaceuticals Inc. (Vanda) (Nasdaq: VNDA) today announced that it has received a decision letter from the U.S. Food and Drug Administration's (FDA) Center for Drug Evaluation and Research (CDER) concluding that the supplemental New Drug Application (sNDA) for HETLIOZ® (tasimelteon) for the treatment of jet lag disorder cannot be approved in its c...
Vanda Pharmaceuticals is buy-rated following FDA approval of Nereus for motion sickness, with shares offering upside into 2026 catalysts. VNDA's Q3 saw strong Fanapt sales growth, but higher R&D and SG&A drove a $22.6M net loss, reflecting a strategic investment phase. The company maintains a robust $293.8M cash position, is guiding to $210–230M in 2025 revenue, and has ample runway to fund mul...
The FDA approved Vanda's drug Nereus to prevent motion-induced vomiting, opening a potential path into treating nausea linked to GLP-1 weight-loss drugs.
Vanda Pharmaceuticals said on Tuesday its drug for the prevention of motion-induced vomiting was approved by the U.S. health regulator, becoming the first treatment for the condition to receive the nod in more than 40 years.
WASHINGTON, Dec. 30, 2025 /PRNewswire/ -- Vanda Pharmaceuticals Inc. (Vanda) (Nasdaq: VNDA) today announced that the U.S. Food and Drug Administration (FDA) has approved NEREUS™ (tradipitant), an oral neurokinin-1 (NK-1) receptor antagonist, for the prevention of vomiting induced by motion. This approval marks the first new pharmacologic treatment in motion sickness in over four decades, repres...
Kostenlos registrieren
aktien.guide ist das Tool zum einfachen Finden, Analysieren und Beobachten von Aktien. Lerne von erfolgreichen Investoren und triff fundierte Anlageentscheidungen. Wir machen Dich zum selbstbestimmten Investor.