Taysha Gene Therapies Inc Aktie News
Taysha Gene Therapies Inc Aktie News

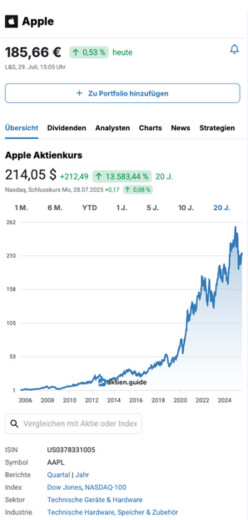
4,66 $
0,00 %
heute
Nasdaq,
12. Februar, 19:48 Uhr
Taysha Gene Therapies Inc Aktie News
DALLAS, Feb. 06, 2026 (GLOBE NEWSWIRE) -- Taysha Gene Therapies, Inc. (Nasdaq: TSHA) (Taysha or the Company), a clinical-stage biotechnology company focused on advancing adeno-associated virus (AAV)-based gene therapies for severe monogenic diseases of the central nervous system (CNS), today announced that, on February 2, 2026, the Compensation Committee of Taysha's Board of Directors granted t...
First patient dosed in REVEAL pivotal trial evaluating TSHA-102 (N=15, aged 6 to
DALLAS, Dec. 05, 2025 (GLOBE NEWSWIRE) -- Taysha Gene Therapies, Inc. (Nasdaq: TSHA) (Taysha or the Company), a clinical-stage biotechnology company focused on advancing adeno-associated virus (AAV)-based gene therapies for severe monogenic diseases of the central nervous system (CNS), today announced that, on December 3, 2025, the Compensation Committee of Taysha's Board of Directors granted t...
Taysha Gene Therapies, Inc. ( TSHA ) Q3 2025 Earnings Call November 4, 2025 8:30 AM EST Company Participants Hayleigh Collins - Director of Corporate Communications & Investor Relations Sean Nolan - CEO & Chairman Sukumar Nagendran - President, Head of Research & Development and Director Kamran Alam - CFO & Corporate Secretary Conference Call Participants Kristen Kluska - Cantor Fitzgerald & Co...
TSHA-102 granted Breakthrough Therapy designation by FDA Finalized FDA alignment on REVEAL pivotal trial protocol and SAP, including 6-month interim analysis that may expedite BLA submission, which was enabled by the rigorous developmental milestone evaluation in Part A REVEAL Phase 1/2 trials showing an unprecedented response rate Dosing of first patient in REVEAL pivotal trial scheduled for Q...
DALLAS, Oct. 28, 2025 (GLOBE NEWSWIRE) -- Taysha Gene Therapies, Inc. (Nasdaq: TSHA) (Taysha or the Company), a clinical-stage biotechnology company focused on advancing adeno-associated virus (AAV)-based gene therapies for severe monogenic diseases of the central nervous system (CNS), today announced that it will report its financial results for the third quarter ended September 30, 2025, and ...
The 2022 Option Agreement between Astellas and Taysha has expired Regaining full rights to TSHA-102 Rett syndrome program enables Taysha to focus on driving its long-term value with full strategic flexibility and optionality TSHA-102 has demonstrated compelling Part A REVEAL Phase 1/2 safety and efficacy data, received FDA Breakthrough Therapy designation and is advancing with a clear path to p...
Previously disclosed 100% response rate across all 10 patients in Part A for pivotal trial primary endpoint of gain/regain of ≥ one natural history defined developmental milestone assessed via video-evidenced review by independent central raters New supplemental analysis of validated, structured efficacy scales provides supportive evidence of additional functional gains, with 100% of patients d...
Kostenlos registrieren
aktien.guide ist das Tool zum einfachen Finden, Analysieren und Beobachten von Aktien. Lerne von erfolgreichen Investoren und triff fundierte Anlageentscheidungen. Wir machen Dich zum selbstbestimmten Investor.