Lexeo Therapeutics Aktie News
Lexeo Therapeutics Aktie News

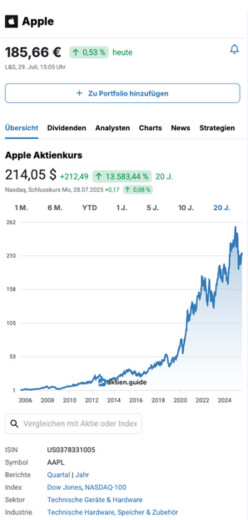
7,39 $
1,47 %
vorgestern
Nasdaq,
16. Januar, 22:00 Uhr
Industrie
Lexeo Therapeutics Aktie News
Neutral
Seeking Alpha
●
4 Tage alt
Lexeo Therapeutics, Inc. (LXEO) Presents at 44th Annual J.P. Morgan Healthcare Conference Transcript
Lexeo Therapeutics, Inc. (LXEO) Presents at 44th Annual J.P. Morgan Healthcare Conference Transcript
Neutral
Seeking Alpha
●
6 Tage alt
Lexeo Therapeutics, Inc. (LXEO) Discusses Preliminary Data From Phase I/II Trial of LX2020 for PKP2-Associated Arrhythmogenic Cardiomyopathy Transcript
Neutral
GlobeNewsWire
●
6 Tage alt
LX2020 generally well tolerated across ten participants with no clinically significant complement activation LX2020 transduction, transcription, and increased protein expression observed across participants with dose-dependent response; mean increase in PKP2 protein of 93% in low-dose cohort and 162% in high-dose cohorts Arrhythmia burden stabilized or improved in majority of participants with...
Neutral
GlobeNewsWire
●
27 Tage alt
NEW YORK, Dec. 22, 2025 (GLOBE NEWSWIRE) -- Lexeo Therapeutics, Inc. (Nasdaq: LXEO), a clinical stage genetic medicine company dedicated to pioneering novel treatments for cardiovascular diseases, today announced that R. Nolan Townsend, Chief Executive Officer of Lexeo Therapeutics, will present at the 44th Annual J.P.
Neutral
Seeking Alpha
●
etwa ein Monat alt
Lexeo Therapeutics, Inc. (LXEO) Clinician Perspectives on Holistic Management of PKP2-Associated Arrhythmogenic Cardiomyopathy Transcript
Neutral
GlobeNewsWire
●
etwa 2 Monate alt
NEW YORK, Dec. 04, 2025 (GLOBE NEWSWIRE) -- Lexeo Therapeutics, Inc. (Nasdaq: LXEO), a clinical stage genetic medicine company dedicated to pioneering novel treatments for cardiovascular diseases, today announced plans to host a virtual event, “A Clinician's Perspective: Holistic Approach to Managing PKP2-Associated Arrhythmogenic Cardiomyopathy,” at the 22nd Global Cardiovascular Clinical Tria...
Positiv
Seeking Alpha
●
2 Monate alt
Lexeo Therapeutics, Inc. has surged after FDA alignment on a pivotal trial for LX2006, targeting Friedreich's ataxia cardiomyopathy. LX2006 shows promising efficacy and safety, with significant LVMI reduction and increased frataxin expression, positioning LXEO as a potential best-in-class therapy. LXEO is well-funded into 2028 after recent financings, and analysts remain bullish, forecasting st...
Neutral
GlobeNewsWire
●
2 Monate alt
FDA open to pooling data from ongoing Phase I/II studies of LX2006 with data from pivotal trial, and to earlier co-primary endpoint assessment, to support a Biologics License Application FDA approved comparability report between LX2006 HEK and Sf9 manufacturing processes in November 2025, endorsing use of Sf9 final commercial manufacturing process to begin dosing patients in upcoming pivotal st...
Kostenlos registrieren
aktien.guide ist das Tool zum einfachen Finden, Analysieren und Beobachten von Aktien. Lerne von erfolgreichen Investoren und triff fundierte Anlageentscheidungen. Wir machen Dich zum selbstbestimmten Investor.