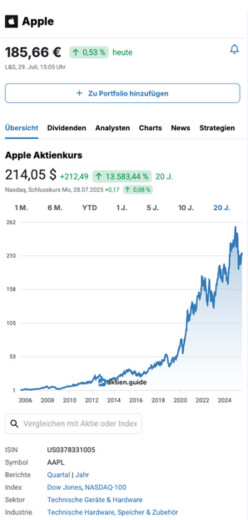
IN8bio Inc Aktie News
IN8bio Inc Aktie News

1,99 $
4,74 %
Nasdaq,
6. Februar, 22:00 Uhr
IN8bio Inc Aktie News
NEW YORK, Feb. 03, 2026 (GLOBE NEWSWIRE) -- IN8bio, Inc. (Nasdaq: INAB), a clinical-stage biopharmaceutical company developing innovative gamma-delta (γδ ) T cell therapies for cancer and autoimmune diseases, today announced that William Ho, CEO and co-founder, will be presenting the following investor and scientific conferences in February. Conference participation details are as follows: Nobl...
NEW YORK, Jan. 12, 2026 (GLOBE NEWSWIRE) -- IN8bio, Inc. (Nasdaq: INAB), a clinical-stage biopharmaceutical company developing innovative gamma-delta (γδ ) T cell therapies for cancer and autoimmune diseases, reported updated clinical data from its INB-200 Phase 1 and INB-400 Phase 2 trials in newly diagnosed glioblastoma (GBM). The prior results were presented at the 2025 Society for Neuro-Onc...
$20.1 million upfront with a milestone-driven additional $20.1 million to advance INB-619, IN8bio's novel gamma-delta (“γδ”) T cell engager through an Investigational New Drug (“IND”) application Financing led by Coastlands Capital with participation from new and existing biotechnology investors Initial proceeds extend cash runway into the first half of 2027 NEW YORK, Dec. 19, 2025 (GLOBE NEWSW...
Presented new preclinical data on INB-619, a novel gamma-delta (γδ) T cell engager (TCE), demonstrating equivalent potency comparable to FDA-approved commercial products with minimal adverse cytokine release, highlighting its potential to achieve deep B cell depletion with an improved safety profile Expanded Phase 1 INB-100 trial to The James Comprehensive Cancer Center at The Ohio State Univer...
IN8bio Expands INB-100 Phase 1 Clinical Trial with Addition of The Ohio State University as New Site
NEW YORK, Oct. 29, 2025 (GLOBE NEWSWIRE) -- IN8bio, Inc. (Nasdaq: INAB), a clinical-stage biopharmaceutical company developing innovative gamma-delta T cell therapies for cancer and autoimmune diseases, today announced that The Ohio State University has been added as a new clinical site in the ongoing Phase 1 trial of the Company's INB-100, a donor-derived allogeneic gamma-delta T cell therapy ...
INB-619 demonstrated equivalent efficacy to the FDA approved, commercial T Cell Engagers (TCE) compounds, blinatumomab and mosunetuzumab, with minimal adverse cytokine release, highlighting a targeted approach that potentially allows for safer deep B cell depletion. INB-619 is the first pan-gamma delta (γδ) T cell engager designed to significantly expand multiple γδ T cell subsets for efficient...
Kostenlos registrieren
aktien.guide ist das Tool zum einfachen Finden, Analysieren und Beobachten von Aktien. Lerne von erfolgreichen Investoren und triff fundierte Anlageentscheidungen. Wir machen Dich zum selbstbestimmten Investor.