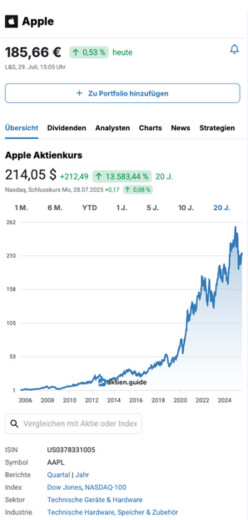
GH Research PLC Aktie News
Biotech stock GH Research catapulted Monday after the FDA gave it the go-ahead to resume testing its depression drug.
GH001 cleared by FDA for U.S. clinical investigation, enabling U.S. subject enrollment Company to seek FDA alignment on global Phase 3 program replicating Phase 2b design Phase 3 initiation targeted for 2026 DUBLIN, Jan. 05, 2026 (GLOBE NEWSWIRE) -- GH Research PLC (Nasdaq: GHRS), a clinical-stage biopharmaceutical company dedicated to transforming the lives of patients by developing a practice...
GH Research is advancing GH001, an inhaled mebufotenin therapy for treatment-resistant depression, with a critical FDA IND update imminent. GH001 demonstrated robust Phase 2b efficacy—a 15.5-point MADRS reduction at Day 8 and 73% six-month remission—with infrequent dosing and strong safety. GHRS maintains a disciplined cost structure, $293.9M in cash, and a projected runway exceeding five years...
DUBLIN, Jan. 02, 2026 (GLOBE NEWSWIRE) -- GH Research PLC (Nasdaq: GHRS), a clinical-stage biopharmaceutical company dedicated to transforming the lives of patients by developing a practice-changing treatment in depression, today reported that it will provide an update on the status of its Investigational New Drug Application (IND) for GH001 with the U.S. Food and Drug Administration (FDA) and ...
Engagement with FDA on GH001 IND complete response ongoing Full dataset from the Phase 2b clinical trial of GH001 in TRD reported in July 2025 Long-term clinical data on safety and efficacy from Open-Label Extension presented at ECNP in October 2025 Cash, cash equivalents and marketable securities of $293.9 million as of September 30, 2025 DUBLIN, Nov. 06, 2025 (GLOBE NEWSWIRE) -- GH Research P...
DUBLIN, Oct. 09, 2025 (GLOBE NEWSWIRE) -- GH Research PLC (Nasdaq: GHRS), a clinical-stage biopharmaceutical company dedicated to transforming the lives of patients by developing a practice-changing treatment in depression, today announced the acceptance of a Novel Therapies Symposium Presentation at the 38th Annual European College of Neuropsychopharmacology Congress (ECNP) in Amsterdam, the N...
Global pivotal program initiation on track for 2026 Engagement with FDA on GH001 IND complete response ongoing The fully completed Open-Label Extension analysis confirms a 73% remission rate at 6 months with infrequent treatment visits and no psychotherapy Treatment was well tolerated and no treatment related serious adverse events were reported. There was no evidence of treatment-emergent suic...
Kostenlos registrieren
aktien.guide ist das Tool zum einfachen Finden, Analysieren und Beobachten von Aktien. Lerne von erfolgreichen Investoren und triff fundierte Anlageentscheidungen. Wir machen Dich zum selbstbestimmten Investor.