BioAtla Inc Aktie News
BioAtla Inc Aktie News

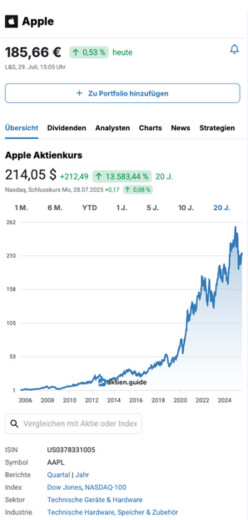
0,21 $
10,78 %
vorgestern
Nasdaq,
6. Februar, 22:00 Uhr
BioAtla Inc Aktie News
SAN DIEGO, Dec. 31, 2025 (GLOBE NEWSWIRE) -- BioAtla, Inc. (Nasdaq: BCAB), a global clinical-stage biotechnology company focused on the development of Conditionally Active Biologic (CAB) antibody therapeutics for the treatment of solid tumors, and GATC Health Corp. (GATC), a tech-bio company leveraging artificial intelligence (AI) to transform drug discovery and development, today announced a S...
SAN DIEGO, Nov. 21, 2025 (GLOBE NEWSWIRE) -- BioAtla, Inc. (Nasdaq: BCAB), a global clinical-stage biotechnology company focused on the development of Conditionally Active Biologic (CAB) antibody therapeutics for the treatment of solid tumors, today announced that it has entered into Pre-paid Advance Agreements with an affiliate of Yorkville Advisors Global (Yorkville) and funds managed by of A...
BioAtla, Inc. ( BCAB ) Q3 2025 Earnings Call November 13, 2025 4:30 PM EST Company Participants Jay Short - Co-Founder, CEO & Chairman Sheri Lydick - Chief Commercial Officer Eric Sievers - Chief Medical Officer Richard Waldron - Senior VP & CFO Conference Call Participants Julie Miller Yu He - H.C. Wainwright & Co, LLC, Research Division Presentation Operator Good day, everyone, and welcome to...
Company achieved FDA alignment on Phase 3 Oz-V trial for the treatment of 2L+ OPSCC, which will evaluate dual primary endpoints with potential of achieving accelerated approval followed by full approval Company is in advanced stages to finalize a strategic transaction with a potential partner and we remain on track to complete the transaction by year end BA3182 trial in advanced adenocarcinomas...
— Mec-V demonstrates median OS of 21.5 months in patients with treatment-refractory leiomyosarcoma, liposarcoma, and undifferentiated pleomorphic sarcoma compared with approximately 12 months with approved agents
SAN DIEGO, Nov. 05, 2025 (GLOBE NEWSWIRE) -- BioAtla, Inc. (Nasdaq: BCAB), a global clinical-stage biotechnology company focused on the development of Conditionally Active Biologic (CAB) antibody therapeutics for the treatment of solid tumors, today announced that it plans to host a conference call and webcast on Thursday, November 13, 2025 at 4:30 p.m. ET to discuss its financial results for t...
SAN DIEGO, Oct. 23, 2025 (GLOBE NEWSWIRE) -- BioAtla, Inc. (Nasdaq: BCAB), a global clinical-stage biotechnology company focused on the development of Conditionally Active Biologic (CAB) antibody therapeutics for the treatment of solid tumors, today announced presentation of a poster entitled “Targeting HPV E6/E7 Upregulation of the Transmembrane Receptor Tyrosine Kinase ROR2 with the ADC Ozuri...
Adverse events (AEs) generally transient and readily manageable; only 2 cases of cytokine release syndrome (CRS) Prolonged tumor control with increasing doses of BA3182; confirmed partial response (cPR) at 0.6 mg in a patient with intrahepatic cholangiocarcinoma without progression for >6 months Maximally tolerated dose not yet defined; dose escalation continues; currently testing 1.8 mg dose ...
Kostenlos registrieren
aktien.guide ist das Tool zum einfachen Finden, Analysieren und Beobachten von Aktien. Lerne von erfolgreichen Investoren und triff fundierte Anlageentscheidungen. Wir machen Dich zum selbstbestimmten Investor.