Benitec Biopharma Inc Aktie News
Benitec Biopharma Inc Aktie News

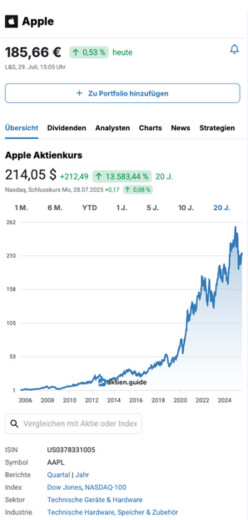
12,12 $
4,03 %
Nasdaq,
6. Februar, 22:00 Uhr
Benitec Biopharma Inc Aktie News
Benitec Biopharma: BB-301's Early OPMD Data And Cash Infusion Justify A Bullish Upgrade
• Patient 1 of Cohort 1 has now completed the 24-month follow-up timepoint, and at month-24 post-treatment Patient 1 continued to demonstrate the powerful disease-modifying effects of BB-301, with deepening improvements in post-swallow residue and total dysphagic symptom burden as compared to the 12-month follow-up timepoint
– Fast Track Designation was granted for BB-301 following U.S. Food and Drug Administration (FDA) review of positive interim clinical study results and proprietary Responder Analysis planned for use in pivotal study for BB-301–
In the past week or so, beneficial owners Carl Icahn and Mario Gabelli made some big bets in Monro Inc.
HAYWARD, Calif., Nov. 06, 2025 (GLOBE NEWSWIRE) -- Benitec Biopharma Inc. (Nasdaq: BNTC) (“Benitec” or the “Company”), a clinical-stage, gene therapy-focused, biotechnology company developing novel genetic medicines based on its proprietary DNA- directed RNA interference (“ddRNAi”) “Silence and Replace” platform, today announced the pricing of its underwritten public offering of 5,930,000 share...
HAYWARD, Calif., Nov. 05, 2025 (GLOBE NEWSWIRE) -- Benitec Biopharma Inc. (Nasdaq: BNTC) (“Benitec” or the “Company”), a clinical-stage, gene therapy-focused, biotechnology company developing novel genetic medicines based on its proprietary DNA- directed RNA interference (“ddRNAi”) “Silence and Replace” platform, today announced that it has commenced an underwritten public offering of its commo...
HAYWARD, Calif., Nov. 03, 2025 (GLOBE NEWSWIRE) -- Benitec Biopharma Inc. (NASDAQ: BNTC) (“Benitec” or “Company”), a clinical-stage, gene therapy-focused, biotechnology company developing novel genetic medicines based on its proprietary “Silence and Replace” DNA-directed RNA interference (“ddRNAi”) platform, today provides positive interim clinical results for the BB-301 Phase 1b/2a Clinical Tr...
Investor webcast to be held on Monday November 3, 2025 at 8:00 am EST Investor webcast to be held on Monday November 3, 2025 at 8:00 am EST
Kostenlos registrieren
aktien.guide ist das Tool zum einfachen Finden, Analysieren und Beobachten von Aktien. Lerne von erfolgreichen Investoren und triff fundierte Anlageentscheidungen. Wir machen Dich zum selbstbestimmten Investor.